Kneat is neat
- Joseph M.
What do you like best about Kneat Gx?
Kneat makes life a whole lot easier in the validation space. No more paper!! Super user friendly and time efficient.



Digitize all your validation processes in one platform, your way, fast.
Trusted by 8 of the world’s top 10 life sciences companies. Proven to deliver data integrity, efficiency, and value on a global scale.
Digitalize any validation process in one platform, your way, without code or compromise.


Unlike some other platforms that focus only on data-centric, form-driven validation — such as Veeva Vault and ValGenesis VLMS — Kneat offered flexibility and configurability.
- Kailash Rathi, Director of Quality Systems & Validation, Recipharm Advanced Bio
Book a demo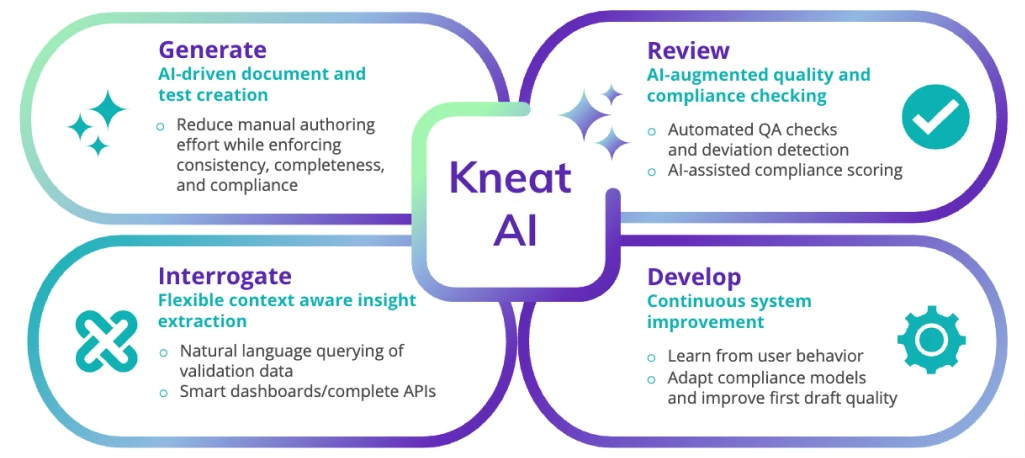
Our approach - GRID
Kneat AI is represented by its ‘GRID’ model, a structured approach that applies AI across the full validation lifecycle. By connecting generation, review, interrogation, and development, GRID ensures AI works with validation data in context, not in isolation.
Because GRID operates inside a validated platform, every AI assisted action remains governed, traceable, and inspection ready. Intelligence is applied where it adds value, while compliance, integrity, and control are maintained at every step.
Customer Ratings
G2
Overall rating by customers via leading software review site, G2.
CSAT Score
97% of our customers rate our customer support as ‘Very Good’ or ‘Excellent’.
PLAN TO RENEW
Customer feedback via industry analyst SoftwareReviews.com.
Any process
Run CSV/CSA, C&Q, Process, Cleaning, Method, Analytical Instrument, Equipment, Utility/Facility, Electronic Logbooks, Drawing and Document Management, Cold Chain, Batch Records— on a single, secure platform. Scale easily across sites and programs to drive deeper ROI as you consolidate tools.

Kneat maps to your workflows, terminology, and risk-based approaches. Teams keep what already works and improve what doesn’t — speeding adoption and reducing change risk.

Enterprise teams deploy quickly, standardize with templates or certified program packs, and expand site by site. Dedicated Customer Success partners bring deep life sciences validation experience to ensure consistency, faster time to value, and scalable adoption.

Automatically generate RTMs, link requirements to testing, and execute online with integrated deviation management. Side-by-side evaluations found Kneat ~40% more efficient for C&Q than a competing paperless solution.

Stage approved documents in a confined space for auditors and external stakeholders — no binders, no scramble, full audit trail. Collaborative review and approval (parallel or sequence) enable remote and faster inspections.
See how Kneat aligns to your SOPs to streamline validation across all processes, reducing cycle times and compliance risk.
Book a demo