Streamline equipment validation with Kneat.
Audits are much less stressful now. We can find what they need instantly and still provide paper copies when requested, which many auditors still like to have and take away.

Cheryl Koit, CSV Manager, Douglas Pharmaceuticals
Go paperless to double productivity. Achieve 85% faster equipment changeovers, cut manual work by 50%, and strengthen data integrity in equipment validation.

Faster protocols
Create standardized protocols to reduce manual effort and drive consistent equipment qualification and validation in pharma, medical devices, and biotechnology across sites and teams.
Digital execution for IQ, OQ, PQ
Replace paper-based execution with IQ OQ PQ software capabilities — capture data in real time and reduce human error during testing.
Data integrity embedded
Support ALCOA++ expectations through controlled workflows that improve accuracy and completeness in equipment qualification and validation records.
Audit readiness by design
Generate a detailed equipment validation system record automatically, including an independent, un-editable audit trail that can be filtered for faster inspection support.
THE SOLUTION
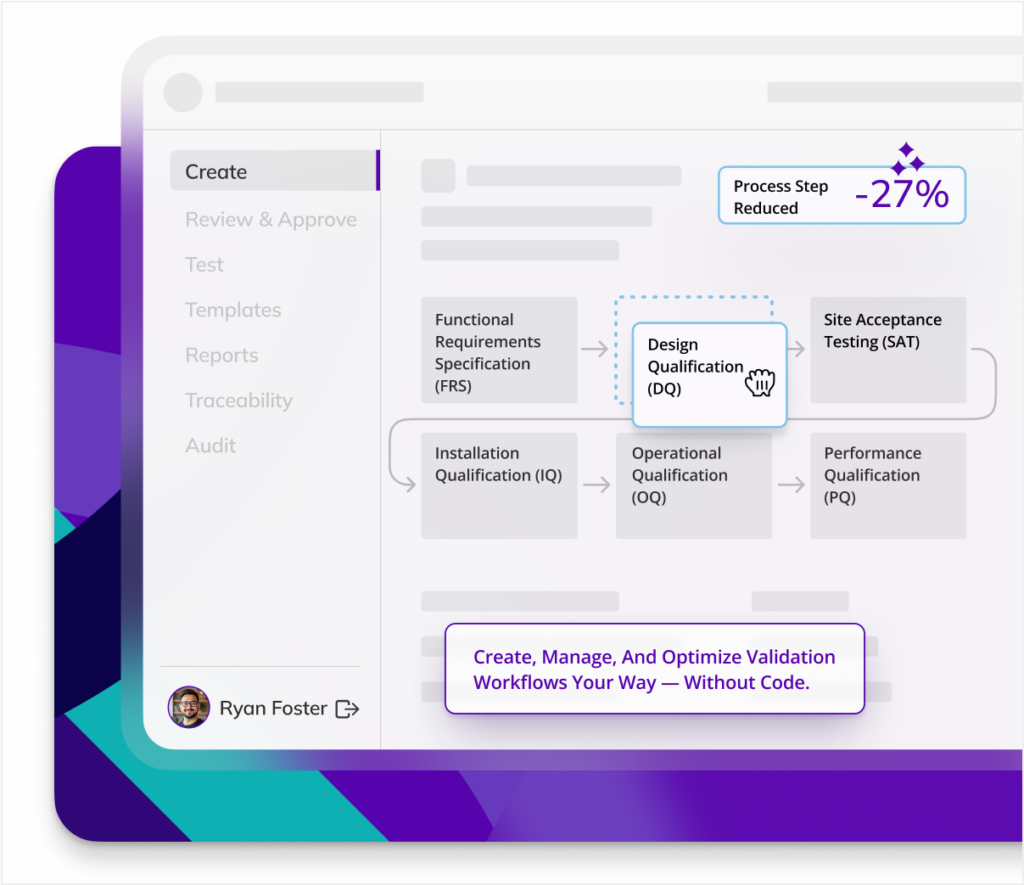
One platform, any process
Manual validation is slow, error-prone, and limits visibility. Kneat streamlines paperless equipment validation by centralizing protocols, execution evidence, review, and approvals in one workflow. Built to scale across CQV — from commissioning through qualification, and validation — Kneat boosts efficiency, strengthens compliance, and reduces risk.
ANY PROCESS
Any process: document or data-centric
Create any validation process, your way — anytime — within one complete solution. Choose a document or data-centric approach and enjoy the flexibility to build and manage your process flow the way you require, whatever your validation methodology.

A real testament to the success of Kneat is when other units, after seeing its capability, request to have their key processes configured on the application. Expansion to other departments and processes is ongoing.
- QA Engineer, Biogen Inc.
Book a demoCustomer Ratings
G2
Overall rating by customers via leading software review site, G2.
CSAT Score
97% of our customers rate our customer support as ‘Very Good’ or ‘Excellent’.
PLAN TO RENEW
Customer feedback via industry analyst SoftwareReviews.com.
See Kneat in action
Book a 30-minute personalized demo of the equipment validation platform trusted by 8 of the top 10 global life science organizations.
Book a demo