
Cut CQV Cycle Times by 40%
Reduce validation cycle times, cut operational risk, and stay audit-ready with Kneat’s digital CQV platform.
Trusted by 8 of the world’s top 10 life sciences companies. Proven to deliver data integrity, efficiency, and value on a global scale.
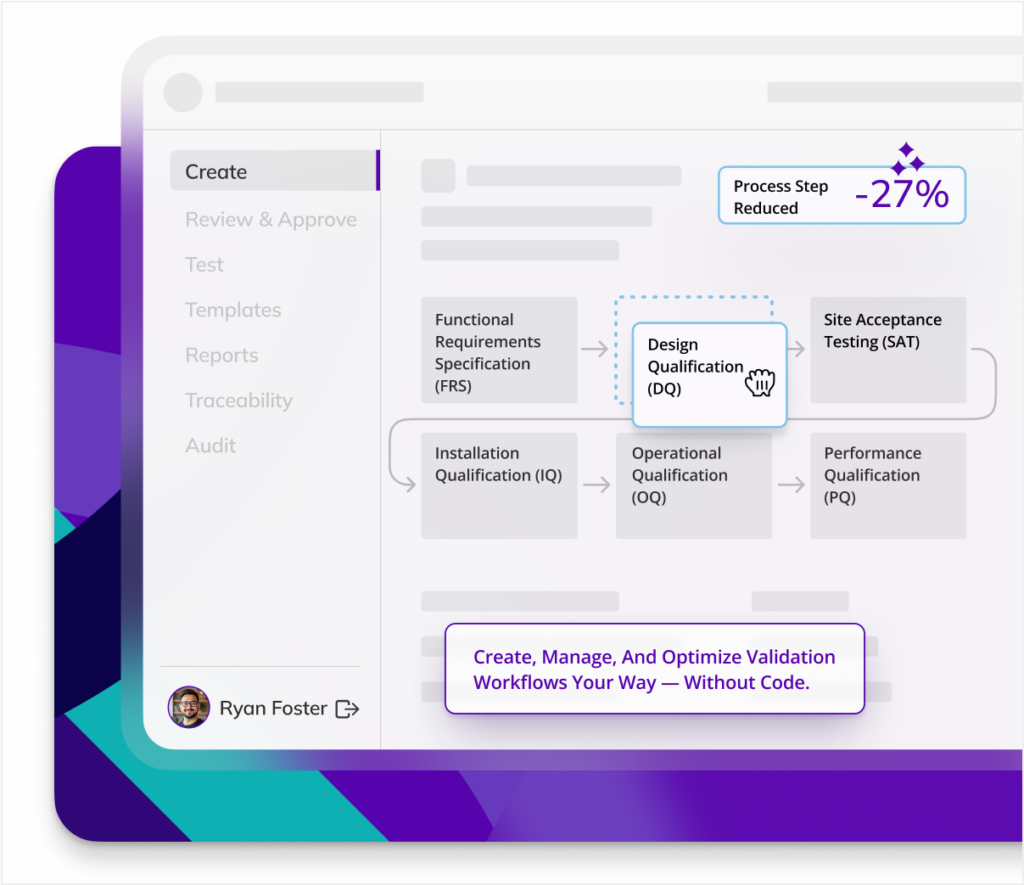
Digitalize any validation process in one platform, your way, without code or compromise.

Efficiency
Reduce cycle time in every step of your validation life cycle, for any process - from protocol creation to review-approval, test execution and more.
Audit readiness
Enjoy peace of mind with complete traceability, instant document retrieval, and audit ‘war-rooms’, ensuring confidence and readiness for inspection anytime.
Data integrity
Ensure unparalled data integrity with secure, time-stamped audit trails, role-based access controls, and automated traceability, enhancing compliance and trust in your validation processes.
Harmonization & governance
Enhance governance through harmonized validation processes across all your sites, ensuring consistent compliance, streamlined oversight, and improved operational control.
Any process
CQV, your way
Kneat centralizes commissioning, qualification and validation in one platform to improve efficiency and compliance while reducing delays and regulatory risk.
- Automate deviations for faster issue resolution and compliance
- Smooth handovers with centralized real-time documentation
- Stay audit-ready with standardized, up-to-date validation processes

We conducted a side-by-side comparison with Kneat and another vendor and found Kneat to be 40% more efficient for C&Q.
- European Lead, Commissioning & Qualification
Customer Ratings
G2
Overall rating by customers via leading software review site, G2.
CSAT Score
97% of our customers rate our customer support as ‘Very Good’ or ‘Excellent’.
PLAN TO RENEW
Customer feedback via industry analyst SoftwareReviews.com.
See Kneat in action
Join our CQV experts for a tailored demonstration focused on your validation challenges and inspection requirements.
Book a demo