Paperless validation
Frustrated with inefficient paper-based validation?
Accelerate production and reduce overhead with Kneat’s
100% digital GxP validation software, purpose-built for
highly regulated companies.
Paperless, powerful
Paper slows any process that relies on it. Digitizing the validation lifecycle accelerates production, improves analysis, enhances compliance, and drives GxP through the entire lifecycle.
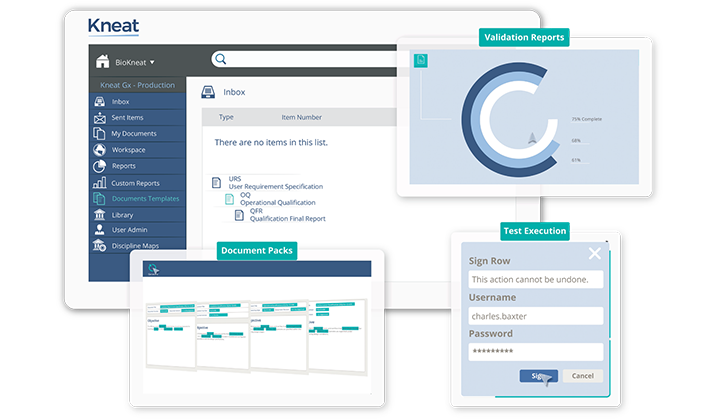
Purpose built, all in one
Author, review, approve, execute testing with integrated exception management, post approve and close out any validation deliverable within one purpose-built, 21 CFR /Annex 11 compliant, web-based platform. Manage GxP and non-GxP documents, drawings, and data in a platform built on a foundation of 50+ years of life sciences industry experience.
Be audit ready, anytime
Kneat gives users unparalleled capability to create, manage, access, and mine validation data for deeper insights and audit preparedness. Kneat enables testers to record results in real time, create and process exceptions, upload evidence, apply electronic signatures, and generate summary reports. Capture any changes, complete with e-signature, time stamp, and reason for change; all while automatically generating a full independent audit trail.
Collaborate in real time
Collaborate with your team from anywhere in the world with Kneat’s secure cloud-based platform. Kneat ensures all documents are always live, up-to-date, and ready for use. No hand-offs, no binder building, no paper retrieval. Faster, smarter collaboration saves time and money while protecting quality and compliance.
Get instant visibility, in-depth
Enjoy at-a-glance insights into every aspect of the validation process across all facilities in real time. Instantly see if a system is validated, due for review, in review, and more. BI dashboards unlock deeper insights including document statuses, user workloads, work processes status updates, across all validation disciplines.

We were able to demonstrate over 50% cycle-time reduction…
and process simplification from fifteen steps to eight…
because of Kneat we minimized the number of systems we used from five to two
Transition
Digitalize Your Paper-Based and Hybrid Validation Program
Whatever the scale of your validation needs, Kneat’s technology and team specialize in guiding the transition from paper to digitized validation quickly and smoothly. Implementation is quick, unintrusive, and fully supported by Kneat.
Easy to start
Whether you’re starting at a single site or rolling out a global strategy, Kneat is built for speedy implementation that seamlessly scales. Add users, disciplines, sites, and more without disrupting operations.
Easy to configure
Kneat is a complete validation solution right out of the box, including any discipline. Enhance the platform with add-ons to suit your needs on an out of the box configurable enterprise platform. Your subject matter experts will be able to quickly configure new, or replicate existing, processes. Quickly re-create your existing processes, documents, and workflows as templates and leverage over and over, across projects and across sites.
Easy to use
Based on designs validation professionals are already familiar with, Kneat’s user interface is designed to become second nature quickly, making the transition to digital validation seamless. If your team can use Windows, they can use Kneat.
Fully supported
Kneat’s experienced support teams will still be there to guide you through process mapping, configuration, training, and deployment to help you transition from paper with ease.

Kneat is at the top of the line as far as paperless validation, DMS system, document storage, archiving…I saw what it can do, how configurable it was, that it allowed us to implement what we wanted from the beginning versus trying to build a system around something that was already existing…we chose it because it was the most innovative product out there
Trusted
Kneat’s paperless GxP validation software is trusted by Eengineers to help them perform their work effectively, releasing them from paper, by Managers to create, maintain, and manage best practice processes, by Quality Directors to oversee quality and satisfy compliance, and by CIOs to help deliver digital transformation.
Applications
For all your validation needs
Use Kneat to digitize and manage any validation, commissioning or qualification process.
A complete solution that can scale to meet all your validation needs across all of your facilities globally.
- Kneat Gx | Audit Readiness
- Document Management
- Drawing Management
- Electronic Logbook Management
- Analytical Instrument Validation
- Utility and Facility Validation
- Process Validation
- Method Validation
- Equipment Validation
- Computer System Validation (CSV)
- Commissioning and Qualification
- Cold Chain Validation
- Cleaning Validation
Value
Protect quality and compliance
while saving time and money
Kneat-powered validation is important for all stakeholders: protecting consumers,
strengthening organizations and satisfying regulators.
Contact
Talk to us
Find out how Kneat can make your validation easier, faster, and smarter.
Start your paperless validation revolution by speaking to our experts.